COVID-19 is now considered endemic, meaning it continues to circulate at predictable levels rather...
Read More
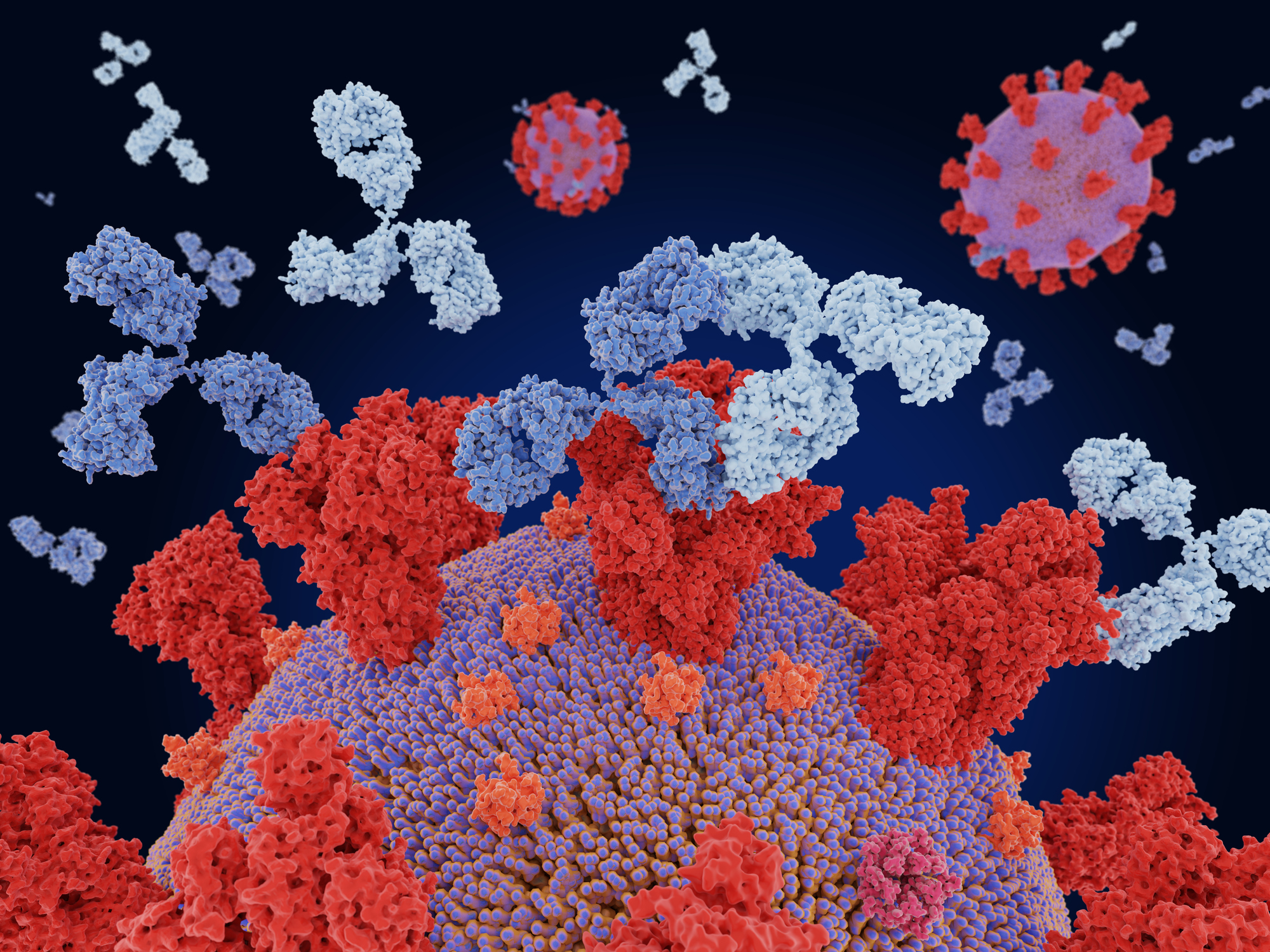
It’s been a long journey since we first found cases of COVID-19 in the United States. But those in the medical field have worked tirelessly from the beginning to research the virus to develop both vaccines and safe treatment options. Now, one of the safest and most effective treatment options available to high-risk COVID-positive patients—monoclonal antibody therapy—is more accessible than ever. However, recent reporting shows that many providers just don’t know where monoclonal antibody treatments are available, let alone how to enroll their patients in treatment.
Monoclonal antibodies are lab-developed molecules that mimic the functions of the immune system targeted toward specific receptors. “COVID-19 infects the body by binding to the ACE2 receptor,” said Craig Sastic, PharmD, B.C.P.S., M.B.A., antimicrobial stewardship pharmacist for Inspira Health. “The monoclonal antibodies prevent this binding from occurring, so the virus is unable to spread throughout the body.”
The treatment is currently available at Inspira Medical Centers Mullica Hill and Vineland via intravenous infusion. “Infusion only takes about 20-30 minutes in our dedicated outpatient infusion rooms,” said Gregory Breen, M.D., a pulmonary and critical care specialist at Inspira Health. “We’ve been offering this to patients for almost a year, and the high-risk patients we’ve treated have tolerated the therapy very well. To date, we’ve observed no significant adverse effects at the time of treatment or afterward.”
This treatment is ideal for patients who have tested positive for COVID-19 and are at high risk for a progression of the virus. “High-risk candidates for the treatment include those who are over the age of 65, are overweight, immunosuppressed or have conditions such as diabetes, cardiovascular disease or lung disease,” said Sastic. There are also groups of people considered high-risk who don’t fit into these set risk factor categories, which means it falls on providers to determine when to recommend this treatment to patients.
It’s important to know that monoclonal antibodies aren’t meant to treat symptoms, but rather to combat the spread of the virus within the body—so it’s most effective when patients come in for treatment within five days of testing positive for COVID-19. Patients are eligible to receive treatment up to ten days after testing positive. “This treatment reduces the risk of hospitalization and potential death, so if a high-risk patient tests positive for COVID-19, they shouldn’t wait to come see us,” said Dr. Breen. “Even patients who are vaccinated, especially if they haven’t gotten a booster and are older or immunocompromised, should come in for treatment.”
Keep in mind that patients cannot get the COVID-19 vaccine or booster for 90 days after infusion.
In addition to treating high-risk patients who test positive for COVID-19, monoclonal antibodies can be used in post-exposure prophylaxis as well. “If someone has been exposed to the virus but hasn’t tested positive yet, they can still get monoclonal antibody treatment,” said Dr. Breen. “For instance, if a high-risk patient lives with someone who tested positive for COVID-19 and they receive this treatment, they have a 70-80 percent less chance of developing the virus themselves.” Patients who are exposed should come in as soon as possible.
During the first year of the pandemic, there was a lot of uncertainty for those who contracted COVID-19. But now, we have treatments that work. “At Inspira, we’ve seen firsthand that the monoclonal antibody treatment works. A very small percentage of patients who receive treatment still require hospitalization. And, even more importantly, the treatment is safe,” said Dr. Breen.
“We’ve seen confirmation of the clinical trials in our own patient population. We’re preventing hospital admissions and we’re preventing mortality,” said Sastic.
As providers, it’s important to communicate the availability and proven effectiveness of monoclonal antibody treatment to your patients who are immunocompromised as they’re coming in for routine visits—instead of after they call you with a positive COVID-19 test result. As more high-risk patients are educated about COVID-19 treatment, more patients will get treated sooner, fewer patients will end up hospitalized and more lives will be saved.
The monoclonal antibody treatment is paid for by the federal government—patients do not need to be insured to receive treatment. If you’d like to refer a patient for monoclonal antibody treatment at Inspira Medical Center Mullica Hill, call 856-853-2025, and Inspira Medical Center Vineland, call 856-641-7557.
COVID-19 is now considered endemic, meaning it continues to circulate at predictable levels rather...
Read More
Respiratory syncytial virus (RSV) is a common respiratory infection that can be serious for infants...
Read More
Zephyr valves are tiny, one-way devices that help people with severe COPD or emphysema breathe...
Read More